“Asia-Pacific IVD Regulatory Affairs Outsourcing Market Size, Share, and Trends Analysis Report—Industry Overview and Forecast to 2029
According to Data Bridge Market Research firms, the Florida Lubricant Solutions Market is set to achieve robust growth, supported by emerging economies and digital transformation. Companies operating in the Florida Lubricant Distribution Market are leveraging advanced technologies to enhance productivity and meet consumer expectations. The demand for customized solutions is rising, further driving expansion in the Lubricants Market in Florida. Leading industry players are focusing on research-backed strategies to strengthen their market position. As competition intensifies, businesses in the Florida Industrial Lubricants Market are utilizing detailed market research reports to understand shifting trends, consumer behavior, and future opportunities in the Florida Automotive Lubricants Market.
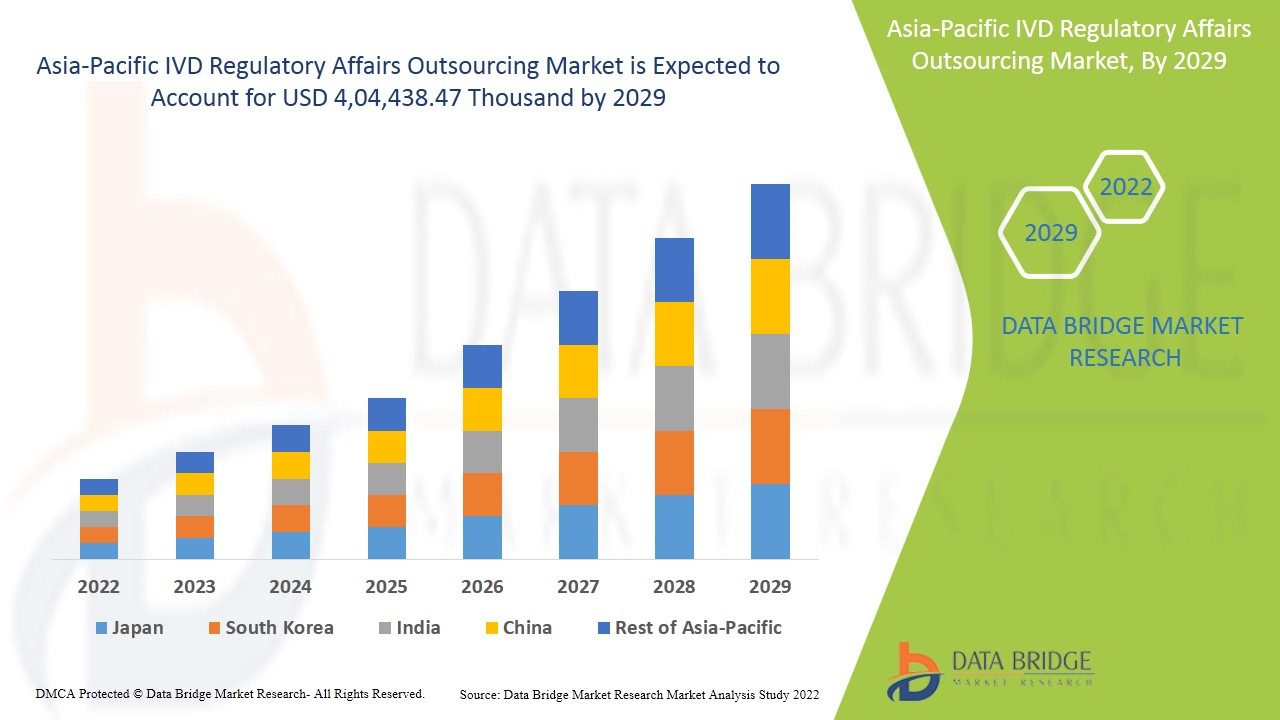
The Asia-Pacific IVD Regulatory Affairs Outsourcing Market is poised for significant growth, with a market outlook highlighting substantial growth potential driven by emerging opportunities in key sectors. This report provides strategic insights, demand dynamics, and revenue projections, offering a comprehensive view of the future landscape, technology disruptions, and adoption trends shaping the industry’s ecosystem evaluation. According to Data Bridge Market Research Asia-Pacific IVD regulatory affairs outsourcing market is expected to gain market growth in the forecast period of 2022 to 2029. Data Bridge Market Research analyses that the market is growing with the CAGR of 14.1% in the forecast period of 2022 to 2029 and expected to reach USD 4, 04,438.47 thousand by 2029.
Leading market research reports highlight the growing use of advanced solutions in the Lubricant Products in Florida Market to improve efficiency and sustainability. Businesses are adapting to regulations, integrating technology, and refining their strategies to stay competitive in the Florida Lubricants Trade Market. The rise of digital transformation has reshaped the Florida Oil and Lubricants Market, pushing companies to invest in automation and smarter business models. With demand rising, companies in the Lubricant Supply Market in Florida are focusing on innovation and customer engagement to stand out. As the industry expands, the Florida Lubricant Chemicals Market presents endless possibilities for businesses ready to embrace change.
Our comprehensive Asia-Pacific IVD Regulatory Affairs Outsourcing Market report is ready with the latest trends, growth opportunities, and strategic analysis. https://www.databridgemarketresearch.com/reports/asia-pacific-ivd-regulatory-affairs-outsourcing-market
**Segments**
The Asia-Pacific IVD regulatory affairs outsourcing market can be segmented based on services, application, and end user. In terms of services, the market can be divided into regulatory writing and publishing, regulatory submissions, clinical trial applications, product registrations, regulatory consulting, and others. Regulatory writing and publishing involve the creation of documents such as technical files, regulatory dossiers, and labeling in compliance with local regulations. Regulatory submissions include the preparation and submission of documents to regulatory authorities for product approval. Clinical trial applications focus on obtaining approvals for conducting clinical trials in different countries. Product registrations involve the registration of medical devices and in-vitro diagnostics with regulatory bodies. Regulatory consulting encompasses advisory services related to compliance, strategy, and risk assessment. The ‘others’ segment may include services such as post-market surveillance and quality management.
On the basis of application, the market can be categorized into infectious diseases, oncology, diabetes, cardiology, autoimmune diseases, nephrology, and others. Each of these applications requires specific regulatory approvals and compliance with regional standards. For instance, infectious disease diagnostics may include tests for tuberculosis, hepatitis, and HIV, among others. Oncology diagnostics cover a range of tests for cancer detection and monitoring. Diabetes diagnostics focus on glucose monitoring and related tests. Cardiology tests include those for heart health and related conditions. Autoimmune disease diagnostics are essential for identifying conditions where the immune system attacks the body’s own tissues. Nephrology diagnostics pertain to kidney-related tests. The ‘others’ category may encompass a variety of additional applications in the IVD sector.
In terms of end users, the market can be segmented into IVD manufacturers, contract research organizations (CROs), academic and research institutes, and others. IVD manufacturers require regulatory support throughout the product lifecycle, from development to commercialization. CROs play a crucial role in conducting clinical trials and may outsource regulatory affairs to specialized service providers. Academic and research institutes engage in diagnostic research and may seek regulatory guidance for compliance with standards. The ‘others’ category could include healthcare facilities, government bodies, and distributors involved in the IVD sector.
**Market Players**
– Company 1
– Company 2
– Company 3
– Company 4
– Company 5
The Asia-Pacific IVD regulatory affairs outsourcing market is witnessing significant growth due to several factors. One of the primary drivers is the increasing complexity of regulatory requirements in the region. As countries in Asia-Pacific enhance their regulations related to medical devices and in-vitro diagnostics, manufacturers are facing challenges in navigating the diverse landscape of compliance. Outsourcing regulatory affairs helps companies ensure adherence to local laws and standards without diverting internal resources from core operations. Moreover, the rising demand for IVD products in the region is driving the need for expedited regulatory approvals, further fueling the outsourcing trend.
Another key growth driver is the expansion of multinational companies into Asia-Pacific markets. As global IVD manufacturers look to penetrate emerging economies in the region, they encounter varying regulatory frameworks that necessitate expert knowledge and support for market entry. Regulatory affairs outsourcing providers offer specialized services tailored to different countries’ requirements, enabling seamless expansion for multinational firms. Additionally, the growing trend of strategic partnerships between outsourcing companies and IVD manufacturers is contributing to market growth. Collaborations allow for knowledge sharing, risk mitigation, and streamlined regulatory processes, benefiting both parties.
Despite the positive growth prospects, the Asia-Pacific IVD regulatory affairs outsourcing market faces certain challenges. One of the major obstacles is the lack of harmonization among regulatory bodies across the region. Varying approval processes, documentation requirements, and timelines pose hurdles for companies seeking to navigate multiple markets simultaneously. Additionally, the evolving nature of regulations and the introduction of new compliance standards create uncertainty for market players. Staying updated with changing regulatory landscapes and ensuring timely submissions remain key challenges for both outsourcing providers and their clients.
In conclusion, the Asia-Pacific IVD regulatory affairs outsourcing market is poised for continued expansion driven by the increasing demand for regulatory expertise, the complexity of regional requirements, and the growth of multinational companies in the sector. Strategic partnerships, technological advancements, and a focus on compliance will be crucial for market players to capitalize on the opportunities presented in the region.
https://www.databridgemarketresearch.com/reports/asia-pacific-ivd-regulatory-affairs-outsourcing-market
The market is highly fragmented, with a mix of global and regional players competing for market share. To Learn More About the Global Trends Impacting the Future of Top 10 Companies in Asia-Pacific IVD Regulatory Affairs Outsourcing Market : https://www.databridgemarketresearch.com/reports/asia-pacific-ivd-regulatory-affairs-outsourcing-market/companies
Key Questions Answered by the Global Asia-Pacific IVD Regulatory Affairs Outsourcing Market Report:
- How will the increasing adoption of Asia-Pacific IVD Regulatory Affairs Outsourcing Market in high-performance computing impact the overall market growth?
- How much is the global Asia-Pacific IVD Regulatory Affairs Outsourcing Market worth? What was the market value in 2024?
- Who are the major players operating in the Asia-Pacific IVD Regulatory Affairs Outsourcing Market? Which companies are the front runners?
- Which recent industry trends can be implemented to generate additional revenue streams?
- How will AI, IoT, and 5G advancements influence the Asia-Pacific IVD Regulatory Affairs Outsourcing Market in the next five years?
- What are the key drivers fueling the growth of the Asia-Pacific IVD Regulatory Affairs Outsourcing Market?
- What are the major challenges and barriers faced by the Asia-Pacific IVD Regulatory Affairs Outsourcing Market?
- How is technological innovation shaping the future of Asia-Pacific IVD Regulatory Affairs Outsourcing Market products?
- What is the impact of government regulations and policies on the Asia-Pacific IVD Regulatory Affairs Outsourcing Market?
- How do supply chain disruptions affect the Asia-Pacific IVD Regulatory Affairs Outsourcing Market?
- What are the regional differences in demand for Asia-Pacific IVD Regulatory Affairs Outsourcing Market products?
- How do revenue streams vary across different sectors of the Asia-Pacific IVD Regulatory Affairs Outsourcing Market?
- What role does technology play in enhancing growth and efficiency in the Asia-Pacific IVD Regulatory Affairs Outsourcing Market?
Browse More Reports:
https://www.databridgemarketresearch.com/reports/global-audio-amplifier-market
https://www.databridgemarketresearch.com/reports/global-breather-membrane-used-in-building-envelope-market
https://www.databridgemarketresearch.com/reports/ireland-radiology-services-market
https://www.databridgemarketresearch.com/reports/north-america-kickboxing-equipment-market
https://www.databridgemarketresearch.com/reports/global-citrus-solvents-market
Data Bridge Market Research:
☎ Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC: +653 1251 982
✉ Email: corporatesales@databridgemarketresearch.com
Tag
Asia-Pacific IVD Regulatory Affairs Outsourcing Market Size, Asia-Pacific IVD Regulatory Affairs Outsourcing Market Share, Asia-Pacific IVD Regulatory Affairs Outsourcing Market Trend, Asia-Pacific IVD Regulatory Affairs Outsourcing Market Analysis, Asia-Pacific IVD Regulatory Affairs Outsourcing Market Report, Asia-Pacific IVD Regulatory Affairs Outsourcing Market Growth, Latest Developments in Asia-Pacific IVD Regulatory Affairs Outsourcing Market, Asia-Pacific IVD Regulatory Affairs Outsourcing Market Industry Analysis, Asia-Pacific IVD Regulatory Affairs Outsourcing Market Key Players, Asia-Pacific IVD Regulatory Affairs Outsourcing Market Demand Analysis“